- ≡ Stegania fluviatilis R.Br., Prodr. Fl. Nov. Holland. 152 (1810)
- ≡ Lomaria fluviatilis (R.Br.) Spreng., Syst. Veg., ed. 16, 4, 64 (1827)
- ≡ Spicanta fluviatilis (R.Br.) Kuntze, Revis. Gen. Pl. 2, 821 (1891)
- ≡ Cranfillia fluviatilis (R.Br.) Gasper & V.A.O.Dittrich in Gasper et al., Phytotaxa 275: 207 (2016)
- = Lomaria rotundifolia Colenso, Tasmanian J. Nat. Sci. 1: 377 (1843) nom. illeg., non Lomaria rotundifolia Blume 1828
- = Lomaria rotundifolia Raoul, Ann. Sci. Nat., Bot. sér. 3, 2: 115 (1844) nom. illeg., non Lomaria rotundifolia Blume 1828
- = Lomaria fluviatilis var. multifida T.Moore, Gard. Chron. n.s.12: 84 (1879)
- = Lomaria fluviatilis var. ramosa Colenso, Trans. & Proc. New Zealand Inst. 20: 225 (1888)
Rhizomes erect, up to 120 mm long (in herbarium material), bearing scales rarely producing stolons. Rhizome scales linear to narrowly ovate, 5–12 mm long, 0.5–1 mm wide, chestnut-brown, bicolorous with paler margins. Fronds strongly dimorphic; sterile fronds 80–900 mm long, forming a prostrate to suberect rosette; fertile fronds on same plant usually equalling or slightly longer than sterile fronds, 80–760 mm long, positioned centrally, held erect. Sterile fronds. Stipes 5–160 mm long, red-brown, densely covered in linear to narrowly ovate chestnut-brown scales with dark brown centres, up to 15 mm long, 1 mm wide. Rachises red-brown proximally and yellow-brown distally, grooved adaxially, densely covered in chestnut-brown scales with dark bases, and white to pale brown hairs 0.1–0.2 mm long. Laminae 65–750 mm long, 12–65 mm wide, pinnatisect distally to 1-pinnate proximally, linear or narrowly elliptic, tapering to a very short, pinnatifid apex, similar colour on both surfaces, herbaceous, bearing hairs and linear scales on both surfaces of the midveins. Pinnae in 8–65 pairs, closely to widely spaced, oblong or elliptic, straight, all of similar length or reducing slightly to the base of the lamina; the longest pinnae 6–35 mm long, 4–15 mm wide, apices obtuse to rounded, margins ± entire to shallowly or irregularly crenate, bases adnate on distal pinnae, sessile on proximal pinnae; the basal pinnae ± opposite. Veins free. Fertile fronds. Stipes 10–250 mm long. Laminae 60–630 mm long, 5–45 mm wide. Pinnae in 9–54 pairs, linear, straight, arising at a narrow angle to the rachis; the longest 4–32 mm long, 1–4 mm wide, replaced by very short sterile segments at the lamina base. Sori and indusia in one row either side of costa, continuous along the length of the pinna.
Blechnum fluviatile is distinguished by its dimorphic fronds, free veins, and sterile laminae that are pinnatisect distally and pinnate proximally. It has a rosette of drooping or prostrate sterile fronds with the fertile fronds held stiffly upright in the centre. The densely scaly stipes and rachises, the narrow sterile fronds with 8–65 pairs of pinnae with obtuse or rounded apices, and the fertile pinnae arising at a narrow angle to the rachis are also characteristic.
One form is known with a normal-looking crown of fronds but with stolons growing from the rhizome (WELT P021208). DNA sequences from the form were identical to plants lacking stolons (Perrie unpub.). Further investigation is needed to determine the extent of stolon formation in B. fluviatile.
Aberrant forms dividing repeatedly at the apex are occasionally found.
North Island: Northland, Auckland, Volcanic Plateau, Gisborne, Taranaki, Southern North Island.
South Island: Western Nelson, Sounds-Nelson, Marlborough, Westland, Canterbury, Otago, Southland, Fiordland.
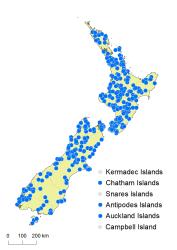
Chatham Islands, Stewart Island, Antipodes Islands, Auckland Islands.
Altitudinal range: 5–1150 m.
Blechnum fluviatile occurs in lowland and montane areas throughout the North Island from Kaitāia to Wellington. It grows from near sea level, up to 1150 m in the Raukūmara Ranges. In the South Island it is widespread in lowland and montane areas, but absent from the drier parts of inland Marlborough, south Canterbury and Otago. It extends from near sea level to 1025 m in the Cobb Valley, north-west Nelson. It also occurs on Stewart Island, the Chatham Islands, Auckland Islands and Antipodes Islands.
Also Sumatra, Borneo, Papua New Guinea and possibly Philippines (Chambers & Farrant 2012), Australia (New South Wales, Victoria, Tasmania).
Blechnum fluviatile is a terrestrial fern found in kauri, podocarp, broadleaved and beech forest, under mānuka and kānuka, and sometimes under Pinus and Salix. It grows in damp, shaded areas on the forest floor, on stream banks and river terraces, in gullies and ditches, near waterfalls, on tracksides, hillsides and roadside banks, in damp grassland, on lake and swamp margins, and on coastal banks.
n = 68 (Brownlie 1954), 2n = 66, 132 (Chambers 1954, 1955). These counts represent diploid and tetraploid populations in New Zealand, but in Australia diploid, triploid, hexaploid and a possible pentaploid count have also been reported (Quinn 1961; Tindale & Roy 2002).
Lomaria fluviatilis var. cristata West, cited by Chambers & Farrant (2001), is a nomen nudum because the epithet “var. cristata” was never used in the original newspaper article (Anonymous 1879 26 April).The type of L. fluviatilis var. multifida T.Moore, said to have been based on the same plant, may have been lost. Apparently 3200 non-British specimens from Thomas Moore’s fern herbarium were given to B in 1888 (Stafleu & Cowan 1981), and may have later been destroyed during World War II. Chambers & Farrant (2001) regarded it as a minor mutant of Blechnum fluviatile.