- Taxon
- Gallery
- ≡ Polypodium dentatum Forssk., Fl. Aegypt.-Arab. 185 (1775)
- ≡ Dryopteris dentata (Forssk.) C.Chr., Kongel. Danske Vidensk. Selsk. Naturvidensk. Math. Afh. 6: 24 (1920)
- ≡ Thelypteris dentata (Forssk.) E.P.St.John, Amer. Fern J. 26: 44 (1936)
- ≡ Cyclosorus dentatus (Forssk.) Ching, Bull. Fan Mem. Inst. Biol. 8: 206 (1938)
- = Polypodium nymphale G.Forst., Fl. Ins. Austr. 81 (1786)
- ≡ Aspidium nymphale (G.Forst.) Schkuhr, 24. Kl. Linn. Pfl.-Syst. 1, 36, t. 34 (1809)
- ≡ Nephrodium nymphale (G.Forst.) Desv., Mém. Soc. Linn. Paris 6: 258 (1827)
- ≡ Dryopteris nymphalis (G.Forst.) Copel., Bernice P. Bishop Mus. Bull. 59: 46 (1929)
- ≡ Cyclosorus nymphalis (G.Forst.) Ching, Bull. Fan Mem. Inst. Biol. 10: 247 (1941)
- ≡ Thelypteris nymphalis (G.Forst.) C.F.Reed, Phytologia 17: 297 (1968)
- = Aspidium molle Sw., J. Bot. (Schrader) 1800(2): 34 (1801) nom. nov. pro Polypodium molle Jacq. 1791 (non Polypodium molle Schreb. 1771)
- ≡ Nephrodium molle (Sw.) R.Br., Prodr. Fl. Nov. Holland. 149 (1810)
- ≡ Dryopteris molle (Sw.) Hieron., Hedwigia 46: 348 (1907)
- = Nephrodium remotum Heward, London J. Bot. 1: 121 (1842)
- = Christella dentata var. caespitosa Holttum, Kew Bull. 41: 518 (1986)
Rhizomes either creeping, up to 150 mm long (in herbarium specimens, but recorded up to 1000 mm in wild populations) with stipes arising 1–10 mm apart in tufts at or near the apex, 2.5–4 mm diameter, or erect and up to 70 mm tall, or very rarely up to 1000 mm tall; bearing scattered scales. Rhizome scales narrowly ovate, 3.5–14 mm long, 0.5–1.2 mm wide, pale or chestnut-brown, entire, with numerous superficial hairs. Fronds 340–1195 mm long, arching upwards. Stipes 45–350 mm long, pale or yellow-brown (or sometimes purple), hairy throughout, scaly proximally. Laminae 1-pinnate to 1-pinnate-pinnatifid, elliptic, narrowed to a pinnatifid apex, 200–945 mm long, 68–350 mm wide, pale to dark green on both surfaces, herbaceous. Scales absent on lamina surfaces; colourless acicular hairs up to 1 mm long abundant on both surfaces and margins; tiny colourless to yellow capitate hairs on abaxial surface. Primary pinnae in 8–30 pairs below pinnatifid apex, widely spaced especially proximally, narrowly ovate or narrowly oblong; the longest at about the middle, sessile, 36–255 mm long, 10–31 mm wide; the basal pair 5–89 mm long, auricled acroscopically. Primary pinnae divided ⅓ to ¾ to the midrib; ultimate segments 5–16 mm long, 3–6 mm wide; apices obtuse to truncate, margins entire and inrolled; sometimes the basal acroscopic segment extended and lobed up to 22 mm long and 11 mm wide. Basal veins on adjacent pinna segments joining, unbranched in each ultimate pinna segment. Sori round, in one row either side of midrib away from pinna margins; indusia reniform, 0.6–1.2 mm diameter, bearing acicular hairs.
Christella dentata is recognised by its herbaceous fronds, primary pinnae divided ⅓ to ¾ to the midrib with obtuse to truncate segments, basal pair of pinnae shortened, veins in adjacent segments joining, indumentum on the abaxial surfaces comprising acicular hairs and tiny capitate hairs but lacking scales, and indusia bearing acicular hairs. The rhizomes are creeping (up to 1 m long recorded in wild populations, de Lange, iNaturalist observation #1463496) to erect (up to 1 m tall, recorded by de Lange, AK 314009), and the fronds arise in tufts at or near the apices, with very short distances between adjacent stipes. This contrasts with the longer-creeping rhizomes with more widely spaced fronds in Cyclosorus interruptus and Thelypteris confluens.
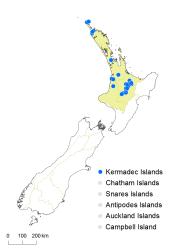
North Island: Northland, Auckland, Volcanic Plateau, Taranaki.
Kermadec Islands.
Altitudinal range: 0–400 m.
Christella dentata has been recorded in lowland sites on the Kermadec Islands (Raoul, Macauley and Cheeseman Islands), from Te Paki (Spirits Bay, Te Huka Bay, Akura Stream, Waitangi Stream), a few localities near Awanui north of Kaitāia, and in thermal regions from Rotorua to Tokaanu. It extends from near sea level to about 300 m on Raoul Island and 400 m near Taupō. It has also been collected from near Kāwhia Harbour, and from Paemako near Piopio, where the status of the populations as indigenous or naturalised is uncertain; neither population is extant. There are populations that are naturalised in a few sites in Auckland and Hamilton, and these may have originated from indigenous plants brought into cultivation at various sites nearby (e.g. AK 305923, Auckland). Others may have originated from cultivated plants with an overseas origin (see below). There is also a specimen in the Armstrong Herbarium (CHR 633413) supposedly collected from Heathcote, Christchurch by J.F. Armstrong in 1863 that might have come from a naturalised plant, although such a record is unlikely at this locality.
Widely distributed in the tropics and subtropics of the Old World from Africa to India, Asia, Australia and most of the islands in the Pacific; extends north to the Azores, Madeira and Crete (Brownsey & Jermy 1973). Naturalised in the Americas (Smith 1971; Holttum 1976) and Hawai‘i (Palmer 2003); Strother & Smith (1970) noted that although common in greenhouses and botanical gardens, it was collected in the New World only twice before 1930, but has since spread very rapidly. Plants in the Neotropics and in Hawai‘i have distinctive purple stipes similar to some naturalised plants in New Zealand (e.g. AK 305922, Auckland) and some plants of uncertain status (e.g. AK 212348, Kāwhia).
On the Kermadec Islands Christella dentata grows in Metrosideros forest, clearings, scrub, open grassland, pumice banks, under overhanging cliffs, disturbed areas, in swampy sites and at the entrance to petrel burrows with Hypolepis dicksonioides. Around Kaitāia and Te Paki it occurs very locally in disturbed and cattle-trampled sites in coastal wetland, on river banks, in streambeds, along roadsides, in drains, under rawiri (Kunzea linearis) forest and in alluvial podocarp forest. In geothermal areas on Raoul Island and the Rotorua/Taupō district it occurs on heated soil, beside hot streams and mud pools, often under mānuka and kānuka in sheltered places or in weedy vegetation. In the western Waikato it was recorded from kahikatea forest. Naturalised plants grow under willows and in disturbed or urban environments.
Indigenous: non-endemic (but naturalised in Auckland and Hamilton and possibly western Waikato).
The species was given a conservation status of ‘At Risk / Naturally Uncommon’ by de Lange et al. (2018). The ‘thermal’ form, not recognised here, was regarded as ‘Nationally Endangered’.
n = 72 (Brownlie 1961, as Cyclosorus nymphalis, Taupō); 2n = 144 (de Lange et al. 2004, Foley’s Bush, Awanui).
Allan (1961) recognised only a single species of Christella in New Zealand (as Thelypteris dentata). Given (1981) suggested that plants from thermal areas might be different to those from around Kaitāia, which he related to "C. dentata of the tropics", but did not elaborate on how they could be distinguished. Brownsey et al. (1985) listed two taxa, C. dentata and C. sp., stating that "two species of Christella may occur in New Zealand, one in thermal areas and the Kermadec Islands, and one in north Auckland". Brownsey & Smith-Dodsworth (1989) distinguished the thermal plant by its shorter rhizome and smaller frond compared to Northland plants but noted that "its taxonomic status and affinities are not yet determined". De Lange et al. (2010) stated that "populations of Christella from geothermally active parts of the North Island and from the crater region of Raoul Island lack the long, creeping rhizome typical of northern New Zealand and most Raoul Island C. dentata, instead producing over time a small, erect trunk. These plants also have narrower, hairier fronds." They concluded that "these plants are not the same as C. dentata, and appear to represent another possibly unnamed variant" but cautioned that further research was still needed.
The taxonomic status of the two forms of C. dentata has been re-evaluated by Brownsey & Perrie (2016). From an analysis of the frond and rhizome morphology, spore size and cytology they found that the only difference between them was the nature of the rhizome and that, in contradiction to Brownsey & Smith-Dodsworth (1989), there were no quantitative frond measurements that could be used to distinguish two taxa. The available evidence strongly suggested that populations in New Zealand are uniformly tetraploid and that there were no differences in investigated DNA sequences between the two forms. Furthermore, they found that the rhizomes, whether creeping or erect, were fundamentally similar in producing tufts of fronds near the apex, rather than fronds that are widely spread along the rhizome (as in families with long-creeping rhizomes such as Dennstaedtiaceae, Hymenophyllaceae or Polypodiaceae). They suggested that the nature of the rhizome may not be of great taxonomic significance, and simply a reflection of different habitats in which the plants are found. Plants in Australia show similar variation. Brownsey & Perrie (2016) concluded that "there is only one rather variable species indigenous to New Zealand, similar to that in Australia, which is correctly identified as Christella dentata.
The picture is complicated by the presence of plants in New Zealand with distinctive purple stipes that are very similar to plants that are naturalised in Hawai‘i and the Neotropics. It is likely that they have naturalised in New Zealand as escapes from cultivated plants originally introduced from overseas. The earliest record is a plant grown from spore collected near Kāwhia in 1987 (AK 212348), but several others have been collected since 1991. Given its history in the Americas, the plant could spread in New Zealand unless carefully controlled.