- ≡ Polypodium rufobarbatum Colenso, Trans. & Proc. New Zealand Inst. 16: 347 (1884)
- ≡ Hypolepis rugosula subsp. rufobarbata (Colenso) Schwartsb. in Schwartsburd & Prado, Acta Bot. Brasil. 28: 217 (2014)
- = Polypodium viscidum Colenso, Tasmanian J. Nat. Sci. 2: 164 (1845) nom. illeg., non Polypodium viscidum Roxb. 1816
Rhizomes long-creeping, 1–2 mm diameter, with stipes arising 10–110 mm apart; bearing red-brown hairs up to 4 mm long. Fronds 115–1180 mm long. Stipes 30–500 mm long, 0.7–3 mm diameter, dark purple-brown to red-brown, bearing a few scattered red-brown non-glandular hairs up to 3 mm long and abundant shorter red-brown glandular hairs up to c. 0.7 mm long. Rachises red-brown proximally, becoming yellow-brown distally, densely covered in red-brown hairs with a few widely spaced non-glandular hairs up to 3 mm long, and many glandular and non-glandular hairs <1 mm long. Laminae 2-pinnate-pinnatifid to 3-pinnate-pinnatifid, narrowly ovate to ovate, tapering to a short pinnatifid apex, 85–830 mm long, 30–340 mm wide, light green on both surfaces, herbaceous; lamina margins and abaxial lamina surfaces bearing red-brown acicular hairs (sometimes pale in juvenile fronds) 0.25–0.5 mm long, interspersed on lamina surfaces with pale glandular and non-glandular hairs; costae bearing some red-brown non-glandular hairs to 1 mm long and many shorter red-brown glandular and non-glandular hairs. Primary pinnae in 10–30 pairs below pinnatifid apex, widely spaced, the proximal pair arising at 30–90° to rachis, winged distally; distal primary pinnae narrowly oblong or narrowly ovate; proximal primary pinnae ovate to narrowly ovate; the longest below the middle, 18–270 mm long, 10–100 mm wide, apices acuminate, bases short-stalked. Secondary pinnae gradually decreasing in length along each primary pinna to the distal end, winged throughout; the longest ovate or narrowly ovate, 6–52 mm long, 3–27 mm wide, apices acute or rarely acuminate in the longest pinnae and obtuse in the shortest, bases short-stalked to sessile or adnate in the smallest fronds. Tertiary pinnae oblong, 1–16 mm long, 1–7 mm wide, apices obtuse, bases adnate, partially divided on larger fronds. Veins ending in apices of ultimate segments. Sori ± round, virtually unprotected; paraphyses absent. Mean spore size 28–32 μm long, 18–21 μm wide; perispores pale, echinate, and reticulate.
Hypolepis rufobarbata is one of three similar species in New Zealand that have glandular hairs, red- or purple-brown stipes and rachises, and lack indusial flaps. Hypolepis rufobarbata is distinguished from H. lactea and H. amaurorhachis by the presence of abundant red-brown acicular hairs on the lamina margin, in contrast to the other two species, which have only colourless glandular or acicular hairs on the margins. Hypolepis lactea is further distinguished by its milky exudate on the adaxial lamina surface at maturity, and by its veins, which are sometimes slightly excurrent. It also has abundant short, colourless, glandular hairs on the rachises and costae, in contrast to H. rufobarbata, which has red-brown glandular or acicular hairs. Hypolepis amaurorhachis is further distinguished by its paler red-brown stipe and rachis becoming green in the distal half.
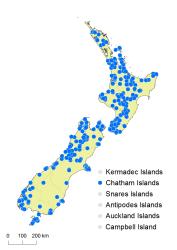
North Island: Northland, Auckland, Volcanic Plateau, Gisborne, Taranaki, Southern North Island.
South Island: Western Nelson, Sounds-Nelson, Marlborough, Westland, Canterbury, Otago, Southland, Fiordland.
Chatham Islands, Stewart Island.
Altitudinal range: 10–1170 m.
Hypolepis rufobarbata is widespread in montane areas of the North Island from near Kaitāia to Wellington, but is scarce on the east coast. It is occasionally found in lowland sites from 50 to 300 m, but occurs most frequently above 300 m, reaching 1100 m in the southern Ruahine Range. In the South Island it is widespread in lowland and montane areas, but is scarce in central Otago and South Canterbury. It mostly occupies a zone from 300 m up to 1000 m but occasionally descends to near sea level in the southern half of the island. It is rare on the Chatham Islands.
Grows on disturbed or boggy soils, swamp margins, rocky ground, clay banks, stream banks, tracksides, and road banks, at the base of uprooted trees, on rotting logs, in humus, on bush margins, in clearings, under scrub, and in forest. It does not tolerate competition from other species and is a coloniser of open ground in damp forested or shaded areas. It grows in podocarp, broadleaved, beech and pine forest, and under mānuka and kānuka.
There is evidence for hybridisation between H. rufobarbata and H. ambigua (AK 170251, CHR 280735, WELT P007817) and H. millefolium (CHR 221720). Hybrids can be recognised by their aborted spores (see Brownsey & Chinnock 1984). The combination H. ambigua × rufobarbata is found commonly where the two species occur together. There is also some morphological evidence that H. rufobarbata and H. lactea hybridise when they occur together (CHR 323893, WELT P011467), but no aborted spores have been found and further work is needed to determine whether such hybrids exist and whether they retain some degree of fertility (Brownsey & Chinnock 1984).
n = 52 (Brownlie 1954, 1957, as H. rugosula; Brownsey & Chinnock 1984).
In New Zealand this species has been widely misidentified by earlier authors as Polypodium rugosulum Labill., P. punctatum Thunb., or combinations based on them.
Hypolepis rufobarbata is morphologically and cytologically distinct from the Australian H. rugosula, which has n = c. 104 (Wakefield 1956; Brownsey & Chinnock 1987). We therefore recognise it here as a distinct species, rather than as a subspecies of H. rugosula as proposed by Schwartsburd & Prado (2014).